BIG NEWS: Drug up for FDA Approval for NF1 Plexiforms
Big news just hit…though it has been expected!
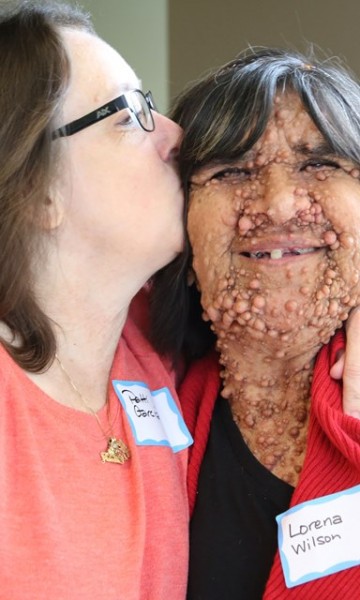
Many of us have been following the success of the drug selumetinib in treating people with neurofibromatosis and plexiform tumors and it was just announced that AstraZeneca and Merck & Co., Inc. has now applied to the US Food and Drug Administration (FDA) for approval of the drug for this use. This applications was given a priority review designation which means that the FDA’s goal is to take action on an the application within 6 months (compared to 10 months under standard review). The application presents selumetinib “as a potential new medicine for pediatric patients aged three years and older with neurofibromatosis type 1 (NF1) and symptomatic, inoperable plexiform neurofibromas (PNs).”
While this is a big step, it’s not the end of the process. If approved, there may still be obstacles in who can get it, insurance coverage, and more. We’ll do our best to keep our community posted!
If you’d like to receive updates from NF Midwest, or if you are in our service region and would like to receive a packet of iNFo, please sign up at www.nfmidwest.org/contact.






.png)






hello
This is great news as I have NF1. I am a 45 years as I want my idenity back. i have very small tumors around my chine line and my trunk area. I have had many surgeries to remove large tumors through out my life time. I would love to have this drug. please keep me posted as I have already signed up to recive emails.
thanks you
Heidi
Hi this is Brindha ,my child has been affected with NF1 i want to receive recent updates regarding NF1
Hi, Brindha! Where are you from? If you’d like to receive email updates you can sign up at http://www.nfmidwest.org/contact.
I will be willing to try it
Do you know if this has been tested on animals?
This drug has been around for awhile, so it is likely that it has been.
This is amazing. I’m about to be on a MEK inhibitor. But I would love to see what this is all about.
At 1st I didn’t understand why I was born with black sport on my body, when I was 18 tumor started to grow us to hate my body I didn’t. Know why it was like this till my father passed away 2010 because of N F 1. He grows tumor on his span after the remove it he passed away a week after, I also have NF1 with tumor all over your body. I wish I didn’t have this tumor always ask my self why me
This would be amazing. I am willing to be a Guinea pig
Me too.
What exactly does this drug do?
This drug has been shown to frequently reduce the size of plexiform fibromas (tumors) in NF1. It has also shown to frequently reduce pain. It is not a treatment for cutaneous neurofibromas or other complications of NF1.
Will it be available for adults with the plexiforms? Or is it just for kids?
We don’t know yet. We’ll keep you posted.
I will be very happy to receive an update from you. I have NF1
In order to get updates you need to provide you info at http://www.nfmidwest.org/contact.